 
The nonpolar amino acids can largely be subdivided into two more specific classes, the aliphaticamino acids and the aromaticamino acids. Colors indicate specific amino acid classes: Hydrophobic – Green and Yellow, Hydrophilic Polar Uncharged – Orange, Hydrophilic Acidic – Blue, Hydrophilic Basic – Rose.Ĭlick Here for a Downloadable Version of the Amino Acid Chart R-groups are indicated by circled/colored portion of each molecule. Each amino acid can be abbreviated using a three letter and a one letter code.įigure 2.2 Structure of the 20 Alpha Amino Acids used in Protein Synthesis. These amino acids are capable of forming full charges and can have ionic interactions. A few amino acids are basic (containing amine functional groups) or acidic (containing carboxylic acid functional groups). Others contain polar uncharged functional groups such as alcohols, amides, and thiols. There are R-groups that predominantly contain carbon and hydrogen and are very nonpolar or hydrophobic. 
The different R-groups have different characteristics based on the nature of atoms incorporated into the functional groups. There are a total of 20 alpha amino acids that are commonly incorporated into protein structures (Figure 2.x). The basic structure of an amino acid is shown below:įigure 2.1 General Structure of an Alpha Amino Acid They differ from one another only at the R-group position. Within living organisms there are 20 amino acids used as protein building blocks. In the diagram below, this group is designated as an R-group. In addition to the amine and the carboxylic acid, the alpha carbon is also attached to a hydrogen and one additional group that can vary in size and length. 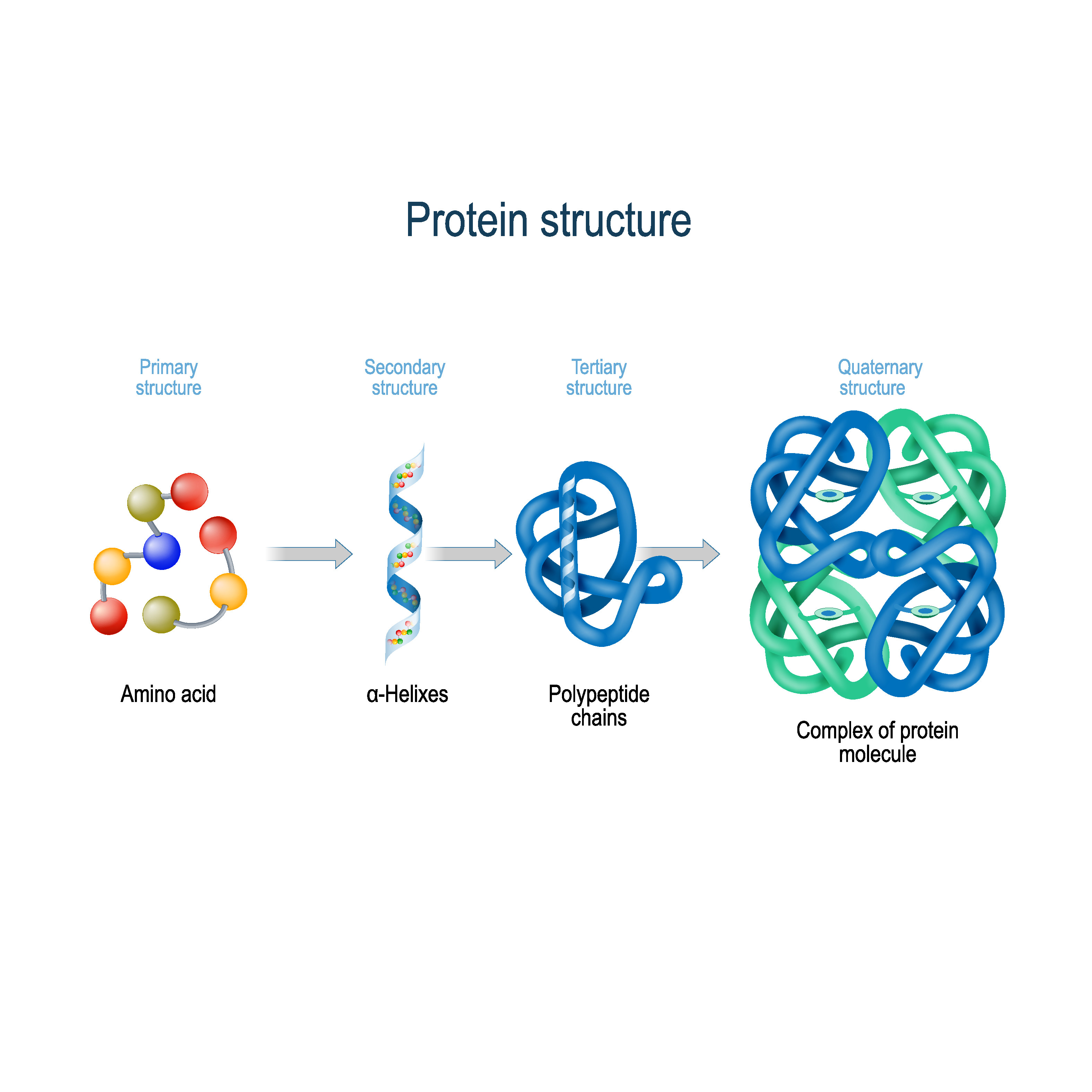 
The alpha designation is used to indicate that these two functional groups are separated from one another by one carbon group. As their name implies they contain a carboxylic acid functional group and an amine functional group. The major building block of proteins are called alpha (α) amino acids. They are all, however, polymers of alpha amino acids, arranged in a linear sequence and connected together by covalent bonds. Their structures, like their functions, vary greatly. Each cell in a living system may contain thousands of different proteins, each with a unique function. Proteins may be structural, regulatory, contractile, or protective they may serve in transport, storage, or membranes or they may be toxins or enzymes. Proteins are one of the most abundant organic molecules in living systems and have the most diverse range of functions of all macromolecules. Primary structure of a protein refers to the sequence of amino acids in the chain.Chapter 2: Protein Structure 2.1 Amino Acid Structure and Properties 2.2 Peptide Bond Formation and Primary Protein Structure 2.3 Secondary Protein Structure 2.4 Supersecondary Structure and Protein MotifsĢ.5 Tertiary and Quaternary Protein Structure 2.6 Protein Folding, Denaturation and Hydrolysis 2.7 References No ads = no money for us = no free stuff for you! Primary Structure Quaternary Structure: interactions between protein subunits that result in the protein being classified as fibrous, globular or conjugated.ĭenaturation refers to the destruction of the three dimensional structure of a protein that results in the loss of biological activity.ĭenaturation of a protein can be caused by:.Tertiary Structure: the interaction between R-groups that causes folding and bending.Secondary Structure: the shape of the protein molecule caused by hydrogen-bonding between -C=O and -N-H groups within the chain.Primary Structure: sequence of amino acids in the chain.There are four types of protein structure: This is known as a condensation reaction, or a condensation polymerisation reaction. Water is eliminated when the amino acids react to form a protein. A polypeptide is made up of many amino acids joined together by peptide bonds (peptide or amide linkages)Ī peptide bond (peptide or amide linkage) is a covalent bond formed between the carbon of the carboxyl group of one amino acid and the nitrogen of the amine group of another amino acid as shown below:.A tetrapeptide is made up of 4 amino acids joined together by peptide bonds (peptide or amide linkages).A tripeptide is made up of 3 amino acids joined together by peptide bonds (peptide or amide linkages).A dipeptide is made up of 2 amino acids joined together by a peptide bond (peptide or amide linkage).You need to become an AUS-e-TUTE Member! Protein Structure and Classification Chemistry Tutorial Key ConceptsĪ protein is a natural polymer, made up of amino acid monomers joined together by peptide bonds (peptide or amide linkages). Want chemistry games, drills, tests and more? Protein Structure Chemistry Tutorial More Free Tutorials Become a Member Members Log‐in Contact Us 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
